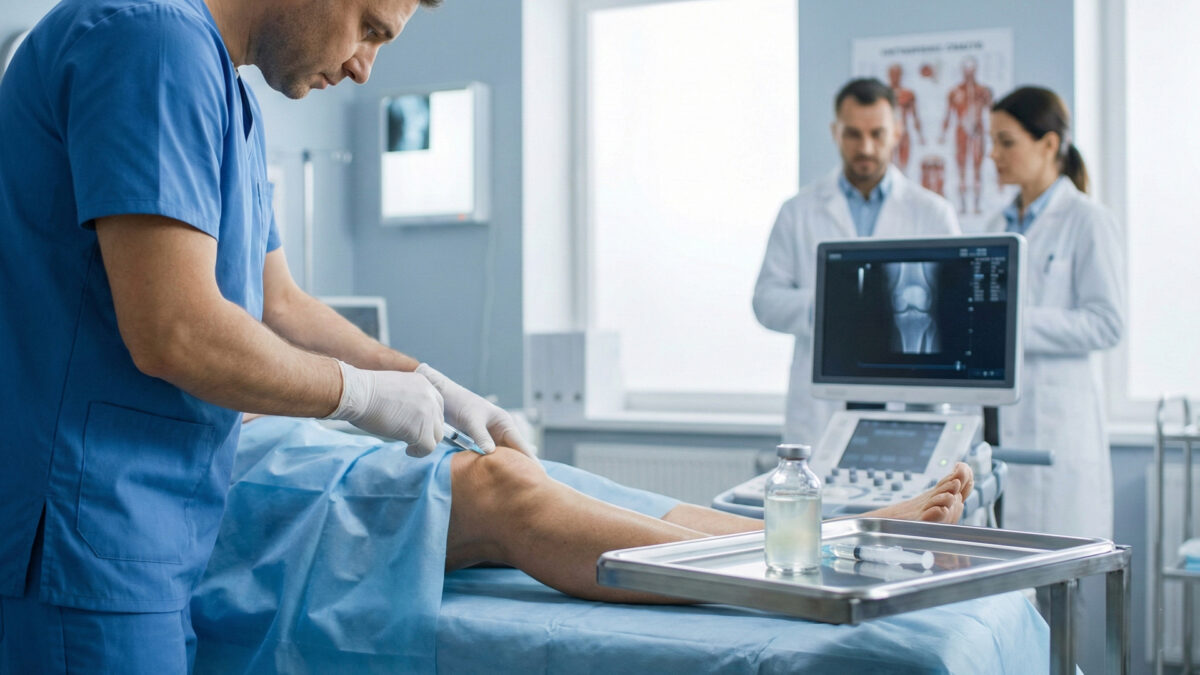
Cipla, in collaboration with Stempeutics Research, has launched a new stem‑cell therapy for knee osteoarthritis called Ciplostem. The therapy is designed to slow joint degeneration and help maintain cartilage health in patients with Grade II or III knee osteoarthritis.
Ciplostem is a ready‑to-use injection containing 25 million cultured bone‑marrow derived mesenchymal stem cells along with hyaluronic acid. Administered directly into the knee joint, it works at the cellular level to reduce inflammation and pain while improving joint function.
Unlike conventional treatments such as painkillers, physiotherapy, or lubricating injections that only relieve symptoms temporarily, Ciplostem targets the root cause of degeneration. The therapy has been developed after extensive clinical research and trials, offering a new regenerative option for patients seeking long-term relief from knee osteoarthritis.
This launch marks a significant step in India’s regenerative medicine landscape, providing a treatment that addresses the underlying joint damage rather than just managing pain.
Also Read: NTT to spend ₹2,400 crore in Bengaluru data‑centre campus